Search
Thermal Reaction Hazards – Your Problem?

“It’s all about Arrhenius: Hot and it grows on you!”
What is this all about, I hear 70% of all our readers ask!?
If you are a chemist, we’re hoping the ‘quip’ above brought at least a groan, or perhaps a slight smile. If you are in the 70% who don’t have a clue what’s going on here and may be a little bit curious about thermal hazards in industry, read on and be aware of one thing; Thermal hazards are often thought of as a problem of the ‘chemical industry’, and this is correct; but beware the industries of food stuffs, detergents, fertilizers, biofuels, metals and even farmers. You can have thermal hazards too. Ignore them at your peril!
The Chemical Industry
We begin with thermal hazards in the chemical industry.
The chemical industry, including pharmaceuticals, plastics, energy…., is an essential component of modern society, providing a wide range of products that are used by everyone. However, chemical manufacturing processes involve a range of hazards, including the release of heat/ energy during chemical reactions or processes, which can lead to thermal hazards. These hazards can pose a significant risk to workers, the environment, and nearby communities and must be understood and managed. In this article, we will provide an overview of thermal hazards in the chemical industry – and in other industries. We will examine how thermal hazards arise, and how they are identified, studied, and indeed, controlled.

What are Thermal Hazards?
Exothermic reactions are chemical reactions that release energy in the form of heat.
Thermal hazards refer to the risks associated with the release of heat/ energy during exothermic reactions, either during processing operations or even simply during substance storage. These hazards, which can arise from intended or unintended chemical reactions, can also release flammable gases or vapors. If gases are generated, the hazards can be particularly serious in the confined space of a chemical reactor (over-pressurization risk), or in the presence of other reactive and/ or flammable chemicals (fire/ explosion risk).
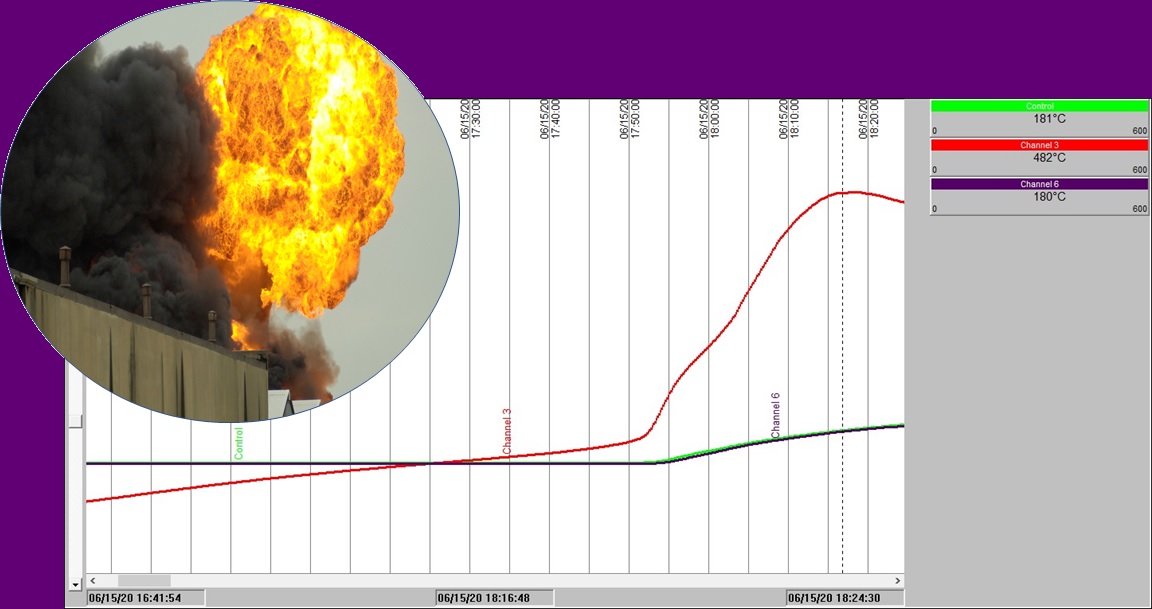
Some chemical reactions can be highly exothermic, releasing a large amount of energy in a short period of time, when the rate of release of heat easily can exceed the rate that heat is lost through natural or forced cooling. It is this energy that can lead to an increase in temperature, gas generation and over-pressure in a reactor or vessel; the classically termed ‘runaway reaction’.
This is where a gentleman of the name Svante August Arrhenius comes in and can offer us an explanation. Arrhenius was a Swedish scientist who won a Nobel Prize for his work in Chemistry. He gave his name to the ‘Arrhenius equation’ (see box) which shows that the rate constant of a chemical reaction increases exponentially with temperature. This means that as the temperature increases in a chemical reaction, the rate of the reaction accelerates. Runaway reaction risk!
One of the best known and now notorious examples of an industrial scale runway reaction happened at a Union Carbide facility in Bhopal, India in 1981 with a methyl isocyanate & water reaction; a reaction that exposed some 500,000 people to the highly toxic gas methyl isocyanate.
In a chemical reactor, heat accumulation depends on the balance between heat generated from the reaction, and heat lost to the surroundings – or more likely to the cooling system installed to moderate reaction temperatures. As a second example of an industrial scale incident closer to home, we refer to the T2 Laboratories runaway reaction incident (Florida in 2007) that left 4 people dead and 14 injured. Reactor cooling failed during the production of methylcyclopentadienyl manganese tricarbonyl (MCMT) reaction. The heat generated could not be removed and a runaway chemical reaction ensued.
How are Thermal Hazards Studied?
To identify and manage thermal hazards, a comprehensive understanding of the chemistry and thermodynamics involved in chemical processes is necessary. Hazard assessments, process design reviews, standard operating procedures and operator training are important components of managing thermal hazards.
Hazard assessments involve identifying and evaluating the hazards associated with a particular process or operation. This includes identifying all potential exothermic reaction and scenarios that could lead to them and assessing the likelihood and consequences of these occurring. In the chemical industry it is usually laboratory testing that provides the information on which hazards studies are based. This can involve study of reaction mechanisms, and kinetics – and measuring parameters such as heats of reaction and decomposition temperatures; it will also involve study of gas generation and rates during the reactions. At Stonehouse our scientists advocate using well established experimental techniques such as ARC (accelerating rate calorimeter), DSC (differential scanning calorimeter) and other specialist equipment to study the reactions under investigation.
The results of the risk assessment are used to develop strategies to manage the risks associated with the process.
Process design reviews are conducted to ensure that the design of the process or equipment can deal with identified reaction hazards and meets regulatory requirements. This includes reviewing the design of the reactor or vessel, the piping and instrumentation diagrams, and the safety systems including reactor relief design. When a reaction does run away, it is essential that the protective systems employed are capable of preventing high pressure build up and explosion risk. Reactor vent design, for example, is a specialist subject with which our chemical engineers are well practiced (DIERS methodology). Process design reviews are important because they can identify potential hazards early in the design phase, allowing for modifications to be made to improve safety.
Establishing safe operating procedures to avoid mischarging of chemicals is also of key importance, but however good the SOP might be, operator training is critical to managing thermal hazards. A well written SOP is of little use if operators find their own ways of doing things! Operators must be trained on the hazards associated with the process, including the potential for thermal hazards. They must also be trained on emergency shutdown procedures, and how to respond to different emergency situations.
Thermal Hazards in other industries
It is important to appreciate that it is not just the chemical industry that has thermal (runaway) hazards! We have written before on the problems of ‘self-heating’ of powders in different industrial settings, where powder decomposition generates heat, usually from oxidation, and where increasing temperature speeds up further heat generation. We have covered the special testing that is possible to establish safe powder drying temperatures in different dryer types and have looked at how basket testing can be used to predict exotherm onset temperatures in hoppers, silos, and packages of different sizes. We have even touched upon the proverbial haystack auto-combustion fire, where the heat source causing ‘auto-combustion of the farmers haystack is likely to originate from biological rather than chemical heat generation processes.
Suffice it to say, with bulked powders too, that when rate of heat generation exceeds the rate of heat loss, temperatures can rise, and the reaction accelerate fast. If left unchecked, this can result in smoldering, fire and sometimes explosion in powder processes following exactly the same ‘Arrhenius equation’ as in the chemical reactions of the chemical industry.
Conclusion – The Stonehouse Approach
Thermal hazards present a significant risk in the chemical and other industries, and from our view and experience, their management requires a comprehensive approach that includes risk assessments, process design reviews, laboratory testing, operator training, and the design and implementation of safety measures such as process control systems, emergency shutdown procedures, and protective equipment. Our Process Safety specialists can work with you to understand your processes and operations, arrange lab testing where necessary and establish/ design suitable protective systems.
As history shows us, the importance of thermal runaway safety in the chemical and other industries should be understood, and companies must appropriately prioritize these hazards to ensure the protection of their workers, the environment, and nearby communities.
By taking a proactive approach to managing thermal hazards, industry can continue to provide the essential materials upon which modern society depends, while minimizing the risks associated with their production.

Get in touch
To learn more about our expertise and services in dust explosion prevention & mitigation, call us at +1 609 455 0001 or email us at [email protected] today.
We also offer tailored virtual and in-company process safety training programs on Dust Explosions, Static Electricity and HAC (Hazardous Area Classification) and more. Find further information here.